If you have been researching weight loss medications in 2026, you have almost certainly encountered two names: Mounjaro and Ozempic. Both are once-weekly injectable medications. Both belong to the GLP-1 drug class. And both have become the most-searched weight loss medications in the world.
But they are not the same drug — and for weight loss specifically, the differences matter more than most people realise.
This pharmacist-written guide covers everything: how each drug works at a molecular level, what the clinical trial data shows in real numbers, how their side effects and drug interactions compare, what they cost in 2026, and which one may be more appropriate depending on your health situation.
What Are Mounjaro and Ozempic?
Before comparing them head-to-head, it is important to understand exactly what each drug is — and crucially, what it is not.
Ozempic (semaglutide) is manufactured by Novo Nordisk and was first approved by the FDA in 2017 for the management of type 2 diabetes in adults. Its active ingredient is semaglutide — the same molecule found in Wegovy (FDA-approved for chronic weight management) and Rybelsus (oral daily tablet for diabetes). Ozempic itself is not FDA-approved for weight loss, though physicians frequently prescribe it off-label for this purpose.
Mounjaro (tirzepatide) is manufactured by Eli Lilly and received FDA approval in 2022 for type 2 diabetes in adults and children aged 10 and older. Its active ingredient is tirzepatide — the same molecule found in Zepbound, which is FDA-approved for chronic weight management. Like Ozempic, Mounjaro is not FDA-approved for weight loss specifically, though it is widely used off-label for this purpose.
Pharmacist’s Perspective — Faryal Faisal, PharmD
Mounjaro and Ozempic are diabetes-approved brands. Zepbound and Wegovy contain the same molecules but are approved for weight loss. The clinical evidence for weight loss comes from all four — because the active ingredient is identical in each pair. When a patient asks me “should I take Mounjaro or Zepbound?” the answer often comes down to which one their insurance covers — not pharmacology.
For guidance on getting a prescription, read: How to Ask Your Doctor for Weight Loss Pills.
The Key Pharmacological Difference: One Receptor vs Two

This is the single most important concept in this entire comparison. The mechanism difference explains nearly every downstream difference — including why one consistently produces more weight loss than the other.
Ozempic (Semaglutide): GLP-1 Receptor Agonist Only
Semaglutide works by mimicking glucagon-like peptide-1 (GLP-1), a hormone your gut naturally releases after eating. By binding to GLP-1 receptors in the pancreas, brain, and gut, semaglutide:
- Stimulates insulin release — only when blood glucose is elevated, which significantly reduces hypoglycaemia risk compared to older diabetes drugs
- Suppresses glucagon — reducing the liver’s release of stored glucose into the bloodstream
- Slows gastric emptying — food leaves the stomach more slowly, producing prolonged fullness and reducing overall caloric intake
- Acts on the brain’s appetite centres — reducing hunger signals and food cravings
Mounjaro (Tirzepatide): Dual GLP-1 + GIP Receptor Agonist
Tirzepatide activates the same GLP-1 receptor as semaglutide — but also activates a second receptor: the glucose-dependent insulinotropic polypeptide (GIP) receptor.
GIP is a gut hormone involved in insulin regulation, fat metabolism, and energy storage in adipose tissue. By activating both receptors simultaneously, tirzepatide produces:
- More powerful appetite suppression than GLP-1 stimulation alone
- Greater improvements in insulin sensitivity
- Larger reductions in body fat, particularly visceral (abdominal) fat
- Faster and larger HbA1c reductions in people with type 2 diabetes
This dual mechanism is the pharmacological reason tirzepatide produces greater weight loss than semaglutide in every major clinical trial conducted to date.
What Does the Clinical Evidence Actually Show?
SURPASS-2: The Direct Head-to-Head Trial
The most important direct comparison is the SURPASS-2 trial, published in the New England Journal of Medicine (Frías et al., 2021). This randomised controlled trial compared tirzepatide at three doses directly against semaglutide 1 mg in 1,879 adults with type 2 diabetes over 40 weeks.
Weight loss results from SURPASS-2:
| Treatment | Average Weight Loss |
|---|---|
| Tirzepatide 5 mg | −7.7 kg (−17 lbs) |
| Tirzepatide 10 mg | −9.3 kg (−20.5 lbs) |
| Tirzepatide 15 mg | −11.4 kg (−25 lbs) |
| Semaglutide 1 mg | −5.9 kg (−13 lbs) |
At the maximum 15 mg tirzepatide dose, patients lost nearly double the weight of those on semaglutide. Tirzepatide also produced significantly greater HbA1c reductions at all three doses.
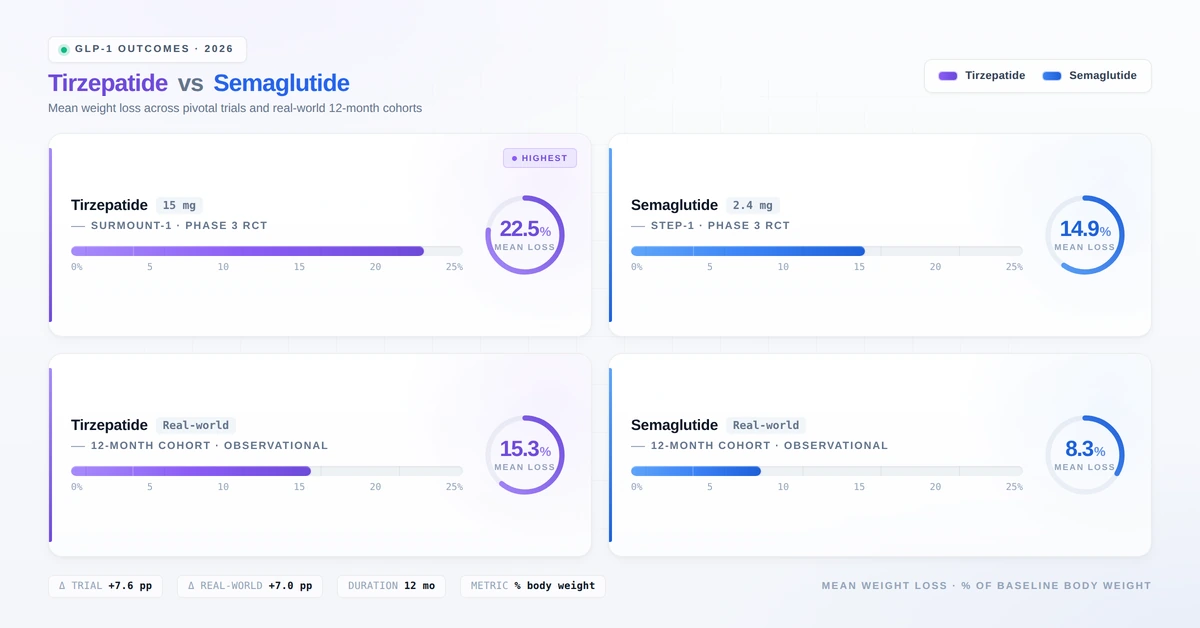
SURMOUNT-1 and STEP-1: Obesity-Specific Trials
SURMOUNT-1 (Tirzepatide for Obesity, NEJM 2022): At the maximum 15 mg dose, participants without diabetes lost an average of 22.5% of their body weight over 72 weeks — approximately 51 lbs for a 225-lb person.
STEP-1 (Semaglutide 2.4 mg for Obesity, NEJM 2021): At the Wegovy dose of 2.4 mg, participants lost an average of 14.9% of their body weight over 68 weeks.
JAMA Internal Medicine: Real-World Comparative Data
A landmark real-world study published in JAMA Internal Medicine by Truveta Research analysed data from over 18,000 patients using propensity score matching.
| Timepoint | Tirzepatide | Semaglutide |
|---|---|---|
| 3 months | −5.9% | −3.6% |
| 6 months | −10.1% | −5.8% |
| 12 months | −15.3% | −8.3% |
Patients on tirzepatide were 1.8× more likely to achieve 5% weight loss, 2.6× more likely to achieve 10%, and 3.2× more likely to achieve 15% weight loss compared to semaglutide.
2025 Meta-Analysis: Pooled Evidence
A systematic review and meta-analysis published in PMC (2025), pooling 2 RCTs and 5 retrospective cohorts, confirmed tirzepatide’s superiority with an overall mean difference of −4.23 kg in favour of tirzepatide (95% CI: 3.22–5.25; p < 0.01). At doses above 10 mg, the difference grew to −6.50 kg.
Dosing Schedules
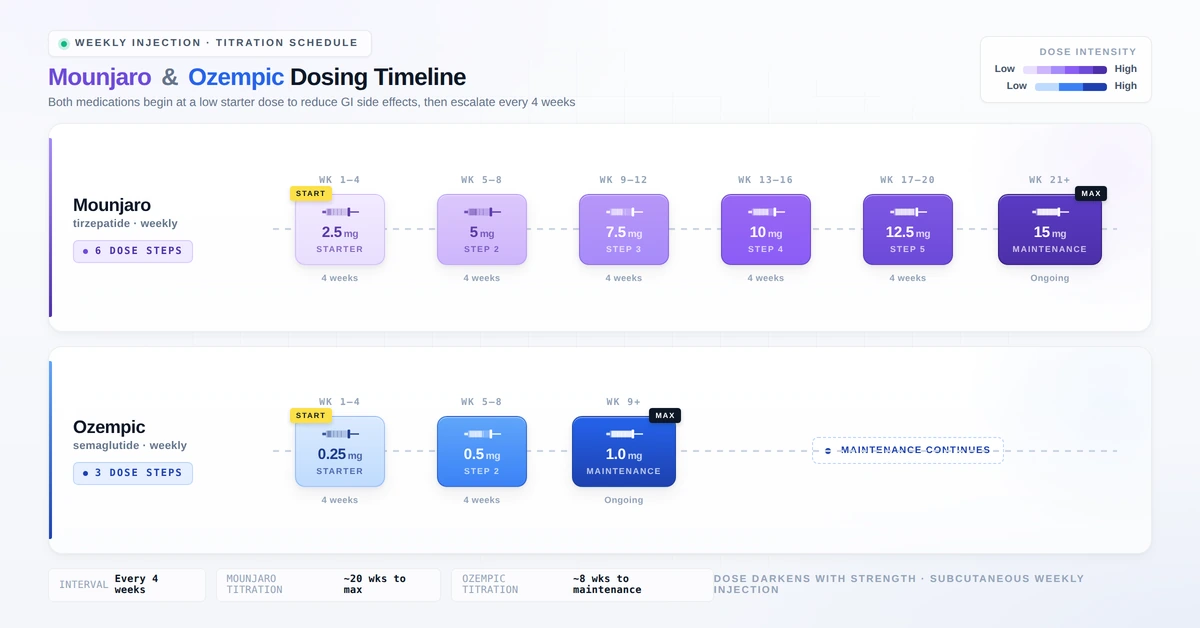
Both medications are administered as once-weekly subcutaneous injections using a pre-filled pen device. Inject into the abdomen, thigh, or upper arm. Rotate injection sites with each dose.
Ozempic (Semaglutide) Dosing
| Phase | Dose | Duration |
|---|---|---|
| Starter dose | 0.25 mg once weekly | 4 weeks |
| Step 1 | 0.5 mg once weekly | 4+ weeks |
| Step 2 (if needed) | 1 mg once weekly | 4+ weeks |
| Maximum dose | 2 mg once weekly | Ongoing |
Missed dose: If within 5 days of scheduled day — take as soon as possible. If more than 5 days — skip and resume regular weekly schedule. Source: Ozempic FDA prescribing information (2025).
Mounjaro (Tirzepatide) Dosing
| Phase | Dose | Duration |
|---|---|---|
| Starter dose | 2.5 mg once weekly | 4 weeks |
| Step 1 | 5 mg once weekly | 4 weeks |
| Step 2 | 7.5 mg once weekly | 4 weeks |
| Step 3 | 10 mg once weekly | 4 weeks |
| Step 4 | 12.5 mg once weekly | 4 weeks |
| Maximum dose | 15 mg once weekly | Ongoing |
Missed dose: If within 4 days — take as soon as possible. If more than 4 days — skip and resume your regular schedule. Source: Mounjaro FDA label (2026).
Pharmacist’s Perspective — Faryal Faisal, PharmD
Neither medication should be stopped abruptly without medical guidance. Weight regain after discontinuing GLP-1 medications is well-documented. Patients often ask me whether they can skip dose escalation to speed up weight loss — the answer is firmly no. The 4-week steps exist specifically to allow your gut to adapt. Rushing the titration is the single most common reason patients abandon these medications because of intolerable nausea.
Side Effects Comparison
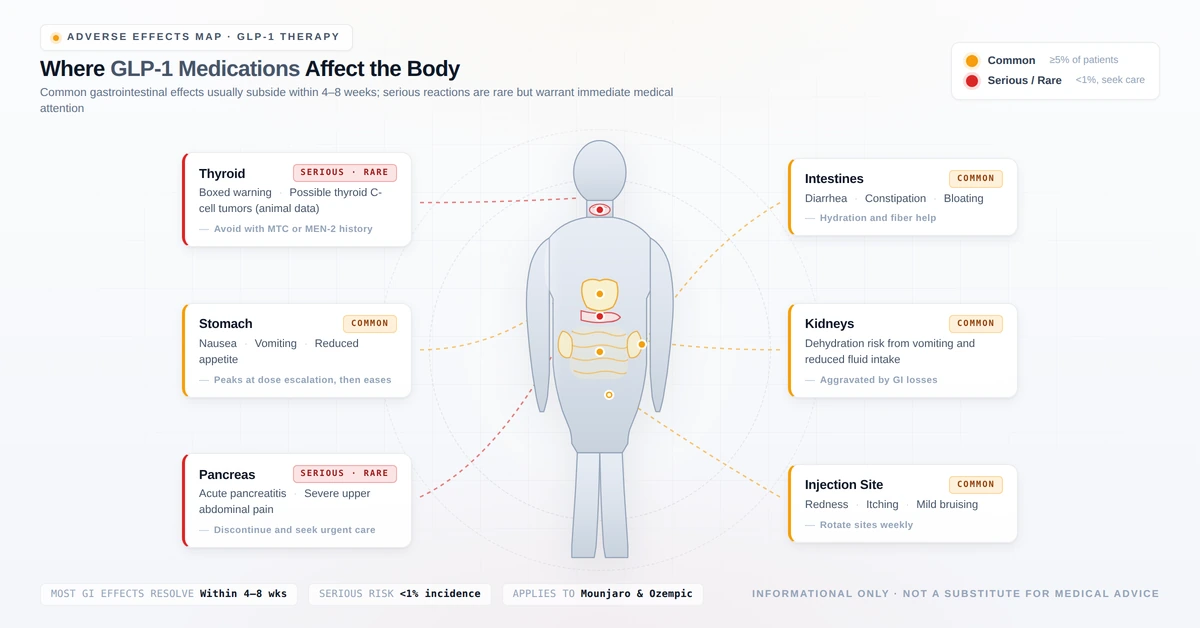
Both medications share a very similar side effect profile. The most common effects are gastrointestinal and most pronounced during dose initiation and escalation.
Common Side Effects
| Side Effect | Ozempic | Mounjaro |
|---|---|---|
| Nausea | Very common (≤20%) | Very common (≤25%) |
| Diarrhoea | Common | Common |
| Vomiting | Common | Common |
| Constipation | Common | Common |
| Decreased appetite | Common | Common |
| Abdominal pain | Common | Common |
| Injection site reactions | Occasional | Occasional |
| Fatigue | Occasional | Occasional |
For most patients, gastrointestinal side effects are mild to moderate and improve significantly within the first 4–8 weeks. Taking the medication with a small meal and avoiding high-fat or spicy foods during the initial weeks helps considerably. For a complete week-by-week timeline of what to expect on semaglutide-based medications, see our pharmacist’s guide to semaglutide side effects week by week.
Serious but Rare Side Effects
1. Thyroid C-Cell Tumours — Boxed Warning (Both Drugs)
Both semaglutide and tirzepatide caused thyroid C-cell tumours in rodent studies. Whether this applies to humans is not established, but both carry a FDA boxed warning and are contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN 2).
Watch for: a lump or swelling in the neck, difficulty swallowing, persistent hoarseness, or shortness of breath.
2. Acute Pancreatitis
Acute pancreatitis has been reported with both medications. Discontinue immediately and seek emergency care if you experience severe, persistent abdominal pain radiating to the back.
3. Hypoglycaemia (Low Blood Sugar)
Risk is significantly elevated when either drug is combined with insulin or sulfonylureas (e.g., glipizide, gliclazide). Your prescriber will likely reduce your insulin or sulfonylurea dose when initiating GLP-1 therapy.
Signs: shakiness, sweating, confusion, dizziness, rapid heartbeat. Treat mild episodes with 15 g of fast-acting carbohydrate.
4. Acute Kidney Injury
Severe dehydration from vomiting or diarrhoea can precipitate acute kidney injury. Patients with pre-existing chronic kidney disease should be closely monitored.
5. Diabetic Retinopathy Complications
Rapid blood glucose improvement — particularly with semaglutide — has been associated with temporary worsening of diabetic retinopathy in patients who already have this condition.
6. Gallbladder Disease
Both drugs are associated with increased rates of cholelithiasis (gallstones) and cholecystitis, partly because rapid weight loss itself promotes gallstone formation.
7. Aspiration Risk During Surgery
Because both drugs delay gastric emptying, patients undergoing elective surgery requiring general anaesthesia should inform their anaesthesiologist. The FDA updated guidance in 2023 recommends considering withholding these medications before procedures.
Drug Interactions: What Pharmacists Watch For
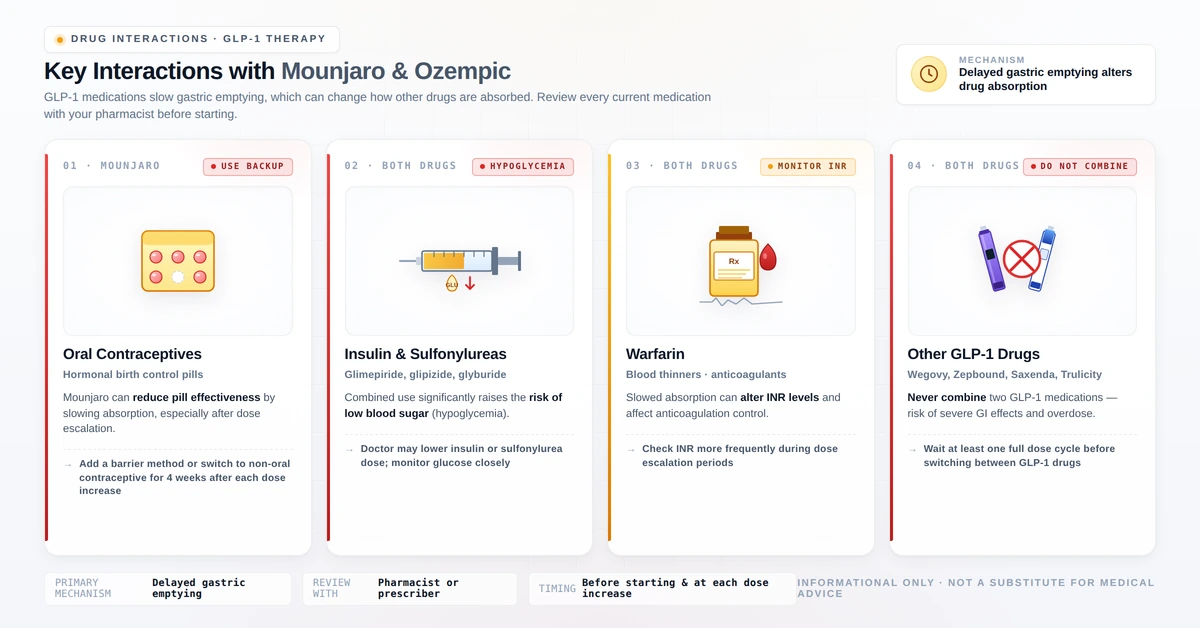
Both medications slow gastric emptying, which alters the absorption timing of oral medications taken concurrently. This is a pharmacokinetically significant interaction affecting several drug categories.
1. Oral Contraceptive Pills — Clinically Critical
Both tirzepatide and semaglutide reduce the peak plasma concentration of oral contraceptive pills by slowing gastric absorption, potentially reducing their effectiveness. Eli Lilly’s prescribing information for Mounjaro explicitly recommends switching to a non-oral contraceptive method for 4 weeks after starting Mounjaro and for 4 weeks after each dose increase.
Recommended alternatives: contraceptive patch, vaginal ring, injectable contraceptive (Depo-Provera), hormonal IUD, copper IUD, or barrier methods.
Pharmacist’s Perspective — Faryal Faisal, PharmD
This is the interaction most commonly missed in clinical practice. In my experience, fewer than half of women starting these medications are properly counselled about the contraceptive impact. If you are of reproductive age and take an oral contraceptive pill, please raise this directly with both your prescriber and your dispensing pharmacist before your first injection.
2. Insulin and Sulfonylureas — Hypoglycaemia Risk
Combining either GLP-1 medication with insulin or sulfonylureas (glipizide, gliclazide, glibenclamide, tolbutamide) significantly increases the risk of hypoglycaemia. Your prescriber will usually reduce the sulfonylurea or insulin dose when initiating GLP-1 therapy. Do not adjust your insulin dose independently.
3. Oral Medications With Narrow Therapeutic Windows
Medications where timing of absorption is critical may be affected:
- Levothyroxine — take consistently on an empty stomach; monitor thyroid function
- Warfarin — INR should be monitored more frequently when starting these medications
- Certain antibiotics — absorption timing may shift; discuss with your pharmacist
4. Other GLP-1 Medications — Never Combine
Do not use Mounjaro and Ozempic simultaneously. Using two GLP-1 medications together dramatically increases the risk of severe hypoglycaemia, pancreatitis, and gastrointestinal complications. If switching between them, follow your prescriber’s guidance on transition timing. See also: our semaglutide dosage guide for switching information.
5. Alcohol
Alcohol does not directly interact pharmacologically with either drug, but it can mask hypoglycaemia symptoms, worsen gastrointestinal side effects, and undermine the dietary discipline needed for optimal weight loss results.
Who Should NOT Take These Medications?
Absolute Contraindications — Do Not Take Either Drug If:
- You have a personal or family history of medullary thyroid carcinoma (MTC)
- You have been diagnosed with Multiple Endocrine Neoplasia type 2 (MEN 2)
- You have had a serious allergic reaction (anaphylaxis, angioedema) to semaglutide, tirzepatide, or any inactive ingredient
- You are pregnant — both drugs should be discontinued at least 2 months before a planned pregnancy. Source: FDA prescribing information
- You are breastfeeding — it is not known whether either drug is excreted in human breast milk
Use With Caution — Discuss With Your Doctor First:
- Pre-existing gastroparesis or severe stomach motility disorders
- History of pancreatitis
- Severe kidney or liver disease
- Pre-existing diabetic retinopathy — particularly relevant for semaglutide
- Scheduled surgery or procedures requiring anaesthesia — discuss withholding the medication with your surgical team
Cost in 2026: What Do These Medications Actually Cost?
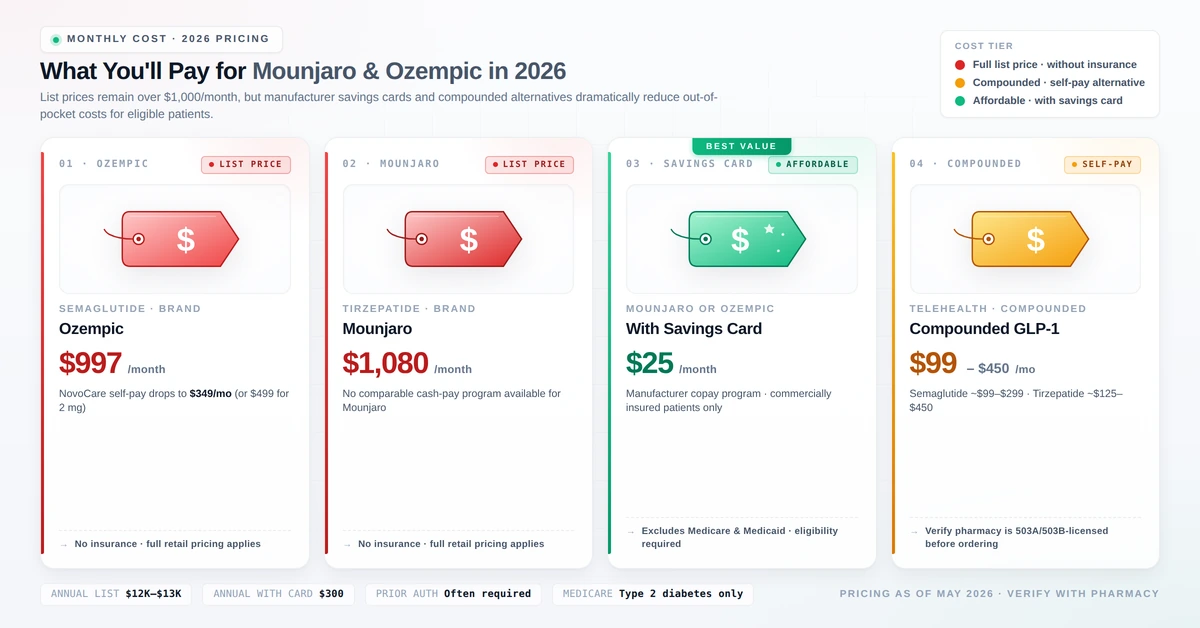
Brand-Name List Prices (Without Insurance, 2026)
| Medication | Active Ingredient | Monthly List Price |
|---|---|---|
| Ozempic | Semaglutide | ~$970–$1,100 |
| Mounjaro | Tirzepatide | ~$1,100–$1,481 |
| Wegovy (weight-loss approved) | Semaglutide | ~$1,300–$1,500 |
| Zepbound (weight-loss approved) | Tirzepatide | ~$1,086 |
Source: GoodRx (2026); Drugs.com (2026).
With Commercial Insurance
Patients with employer-sponsored commercial insurance may pay as little as $25 per month for a 1-, 2-, or 3-month supply using manufacturer savings cards. These cards cannot be used by patients on Medicare, Medicaid, or other government insurance.
Medicare Coverage in 2026
| Drug | Covered for Diabetes | Covered for Weight Loss |
|---|---|---|
| Ozempic | Yes (with prior auth) | No |
| Mounjaro | Yes (with prior auth) | No |
| Wegovy | No | No (except CV risk reduction) |
| Zepbound | No | No (except sleep apnoea) |
The 2026 Medicare Part D out-of-pocket cap is $2,100 per year for covered medications. Source: Wellcare GLP-1 Coverage Guide.
Compounded Alternatives
For patients without coverage, compounded semaglutide and tirzepatide are available through telehealth providers at $99–$450 per month. However, compounded medications are not FDA-approved and quality standards vary. Use only FDA-registered compounding pharmacies and discuss this option carefully with your prescriber. Read more: Weight Loss Shots with B12 — Are They Worth It?
Mounjaro vs Ozempic: Which Is Right for You?
Choose Ozempic (Semaglutide) If:
- You have type 2 diabetes and established cardiovascular disease — semaglutide has a proven cardiovascular benefit (SUSTAIN-6 trial)
- You have pre-existing diabetic retinopathy requiring more gradual glucose reduction
- You have chronic kidney disease — semaglutide has an approved kidney-protective indication
- Your insurance covers semaglutide but not tirzepatide
Choose Mounjaro (Tirzepatide) If:
- Maximum weight loss is your primary goal — the data consistently and significantly favours tirzepatide
- You have type 2 diabetes with persistently poor blood glucose control
- Your insurance or savings programme makes tirzepatide accessible at a similar cost
What Both Drugs Require to Work:
Neither Mounjaro nor Ozempic is a standalone solution. Clinical trials consistently show the best results when these medications are combined with:
- A calorie-conscious, nutrient-dense eating pattern — our 21-Day Anti-Inflammatory Diet Plan pairs well with GLP-1 therapy
- Regular physical activity — especially resistance training to preserve muscle mass during weight loss
- Consistent follow-up with your prescribing healthcare provider
Frequently Asked Questions
Can I take Mounjaro and Ozempic at the same time?
No — never combine these medications. Using two GLP-1 agents simultaneously dramatically increases the risk of severe hypoglycaemia, pancreatitis, and serious gastrointestinal complications. If transitioning between them, follow your prescriber’s guidance on timing.
How long before I see weight loss results?
Most patients notice some weight reduction within 4–8 weeks once therapeutic doses are reached. Significant weight loss (5–10% of body weight) typically occurs within 3–6 months. In real-world data, the average 3-month weight loss was −5.9% for tirzepatide versus −3.6% for semaglutide. Peak results generally appear at 12–18 months.
Will I regain weight if I stop taking these medications?
Yes — this is well-documented for both drugs. Substantial weight regain after discontinuation is common. Both medications are intended as long-term, chronic treatments rather than short-term interventions. Discuss a long-term management plan with your prescriber before starting.
Which drug causes less nausea — Mounjaro or Ozempic?
Both cause similar nausea rates, particularly during dose escalation. Some patients find tirzepatide’s nausea marginally more pronounced at higher doses. Best strategies to reduce nausea: eat smaller meals, avoid high-fat and spicy food, stay upright after eating, stay well hydrated, and do not rush dose escalation.
Is Mounjaro the same as Zepbound?
Yes — both contain tirzepatide at identical doses. The only difference is their FDA-approved indications. Similarly, Ozempic and Wegovy both contain semaglutide. Your prescriber may choose the brand based on your diagnosis and insurance coverage.
Can women on the pill take these medications?
Yes, but with an important precaution. Both medications reduce the effectiveness of oral contraceptive pills due to delayed gastric absorption. Switch to a non-oral contraceptive method for at least 4 weeks after starting either drug and for 4 weeks after any dose increase.
Is Ozempic safe if I have kidney disease?
Ozempic should be used cautiously in advanced kidney disease. However, semaglutide has demonstrated kidney-protective effects in type 2 diabetes patients with chronic kidney disease and is FDA-approved to slow kidney disease progression in this population. Tirzepatide’s kidney data is still emerging.
What about weight loss injections that also contain B12?
These are different products — lipotropic or MIC injections — and work through entirely different mechanisms. Read our full review: Weight Loss Shots with B12 — Are They Worth It?
Key Takeaways
- Tirzepatide is a dual GLP-1/GIP agonist; semaglutide is a GLP-1 agonist only — this explains tirzepatide’s superior weight loss
- Clinical evidence consistently favours Mounjaro: ~double the weight loss at 12 months in real-world data
- Side effects are similar for both — primarily gastrointestinal, worst during dose escalation, improving over time
- Critical interactions: oral contraceptives, insulin, sulfonylureas, and warfarin
- Both are contraindicated in personal/family history of MTC or MEN 2, and during pregnancy
- Brand-name costs exceed $1,000/month without insurance; commercial savings cards can reduce this to $25/month
- Neither drug works optimally without concurrent dietary changes and physical activity
References
- Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385:503–515. https://www.nejm.org/doi/full/10.1056/NEJMoa2107519
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1). N Engl J Med. 2022;387:205–216. https://www.nejm.org/doi/full/10.1056/NEJMoa2206038
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity (STEP-1). N Engl J Med. 2021;384:989–1002. https://www.nejm.org/doi/full/10.1056/NEJMoa2032183
- Real-world comparative effectiveness of tirzepatide vs semaglutide for weight loss. JAMA Intern Med. 2024. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2817610
- Comparative efficacy of tirzepatide vs semaglutide — systematic review and meta-analysis. PMC. 2025. https://pmc.ncbi.nlm.nih.gov/articles/PMC12151102/
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes (SUSTAIN-6). N Engl J Med. 2016;375:1834–1844. https://www.nejm.org/doi/full/10.1056/NEJMoa1901118
- Mounjaro (tirzepatide) Prescribing Information. Eli Lilly. 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2026/215866s009lbl.pdf
- Ozempic (semaglutide) Prescribing Information. Novo Nordisk. 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s035lbl.pdf
- Mando N, et al. Acute pancreatitis caused by tirzepatide. Cureus. 2024;16(12):e76007. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11743417/
- GLP-1 receptor agonists — mechanism overview. StatPearls, NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK551568/
- GoodRx. How much is Mounjaro without insurance? 2026. https://www.goodrx.com/mounjaro/how-much-is-mounjaro-without-insurance
- Drugs.com. Mounjaro cost and insurance 2026. https://www.drugs.com/medical-answers/mounjaro-covered-insurance-medicare-3571888/